.png)
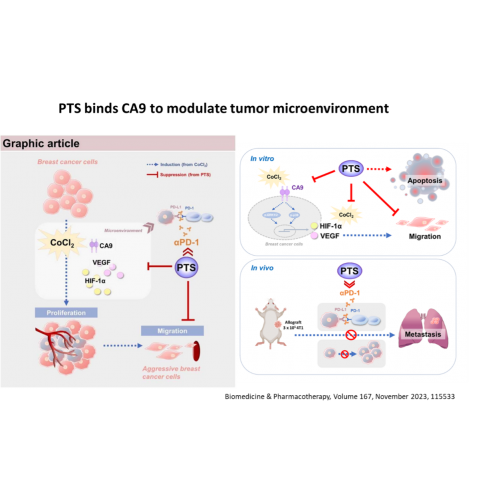
Cancer cells are with more abundant CA9 on the cell membrane than normal cells such as lung cancer, liver cancer, H&N cancer, breast cancer and melanoma, resulting in active PI3K-AKT-HIFα mechanism, causing cancer cell proliferation and acidification of the tumor microenvironment. PTS upregulated the expression of apoptosis-related proteins and downregulated CA9, HIF-1α, and VEGF proteins, possibly through modulation of p38 MAPK and ERK1/2 phosphorylated proteins. PTS can bind to CA9 directly to modulate the tumor microenvironment and exhibiting synergistic effects when combined with immune-checkpoint inhibitor αPD-1 therapy.
.png)
In the 2021 study, it was further confirmed that PTS interferes with cholesterol recycling to the cell membrane, resulting in a decrease in the cholesterol content of the cell membrane of lung cancer cells, which in turn interferes with the stability of the lipid raft structure, resulting in the blockage of downstream cancer cell survival, proliferation and metastasis. At the same time, it also activates autophagy, which eventually leads to apoptosis of cancer cells.
.png)
Left:
Cancer cells (such as lung cancer, liver cancer, breast cancer, head and neck cancer, prostate cancer, etc.) are with more lipid rafts structures on the cell membrane than normal cells, resulting in active AKT-mTOR-p70S6K mechanism, causing cancer cell proliferation.
Right:
When PTS enters cancer cells, PTS causes abnormal cholesterol metabolism pathways in cancer cells, which interferes the stability of the lipid rafts structure, resulting in the blockage of downstream cancer cell survival, proliferation and metastasis pathway (AKT-mTOR-p70S6K) , and eventually leads to apoptosis. Therefore, when PTS enters cells, it has a greater impact on cancer cells than normal cells
.png)
Through the intratumoral injection route, PTS greatly reduces the impact on surrounding normal cells, and eventually destroys tumor cell lysosomes and mitochondria, resulting in apoptosis and necrosis of tumor cells.