.jpg)
Pet Medical Care Division
Preliminary Clinical Trial Case
Canine Left Mandibular Oral Cavity Recurrent Malignant Melanoma
.png)
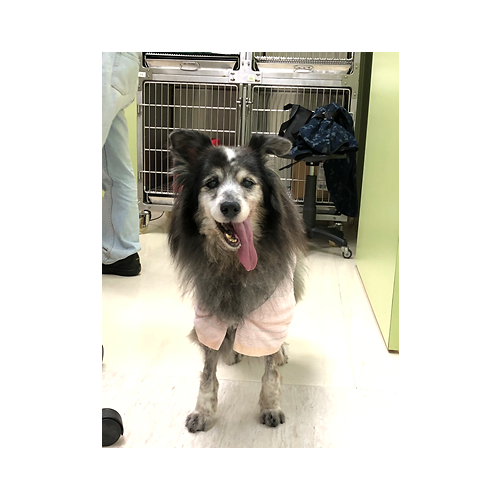
Object Animal:Dog
Breed: Mix
Name:Yao-Yao
Gender:Female
Age:16 years old
Weight:13~14kg
Diagnosis: Recurrent malignant melanoma on left mandibular oral cavity.
Case background:
In 2018, malignant melanoma was found in Yao-Yap’s left mandibular oral cavity. The melanoma has spread over the mandible, so the it was surgically removed. At the same time, the left mandible was also removed. In March 2020, a new tumor about one cubic centimeter was found at the original lesion, which was determined to be malignant melanoma by histopathological biopsy; according to the veterinarian’s evaluation, the tumor was growing rapidly, so it is recommended to use PTS to locally control the tumor first, and then choose a more suitable treatment method to delay the possibility of relapse, depending on the recovery status.
.png)
.png)
.png)
Gongwin Biopharm Co., Ltd. started Pet Drug Project in December 2018, and obtained the approval from the Bureau of Animal and Plant Health Inspection and Quarantine, COA, Executive Yuan (Prevention Inspection First No.1081415339) in November 2019 for the implementation of field trials on dogs; meanwhile, Taipei City Animal Protection Office visited Gongwin Biopharm to complete the inspection and verification on special labels for animal trial production of drug samples (Animal Protection Inspection No. 1086026496); in February 2020, the Bureau of Animal and Plant Health Inspection and Quarantine, COA, Executive Yuan, agreed to expand the field pre-test sites (Prevention Inspection First No 1091400445).
.png)
Yao-Yao received five Intratumoral injection treatments of "animal drug sample trial production of PTS", a total of 7.8 mL.
.png)
Yao-Yao's oral malignant melanoma has completely shrunk and fell off on the 13th day of the preliminary clinical trial treatment course. The residual lesions of about 0.1 cm could be seen at the original lesion. The cell staining result after fine needle aspiration cytology (FNA) showed non-malignant tumor tissue. ; and on the 19th day of the treatment course, the computed tomography scan showed that, during the course of this preliminary clinical trial, there appeared to be no metastasis in other organs; meanwhile, on the day of the computed tomography, the veterinarian removed the residual lesion in order to conduct the histological staining, and the result confirmed that the residual lesion was non-malignant tumor tissue.
.png)
.png)
The Pet Medical Care Division is honored to have gained the trust of the owner, so Gongwin Biopharm can provide more treatment options for pets suffering from tumors. Gongwin Biopharm Pet Medical Care Division will continue to make every effort to contribute to the treatment of dogs suffering from tumors.
.png)
.png)
.png)